Removing Essure Devices
The Entire Device Is Not Visible
The Essure device is 4 cm long. The manufacturer recommends 3 to 8 turns of the outer coil be allowed to project into the uterine cavity to insure adequate placement.
When properly placed approximately 1 to 2 cm of the Essure device should project beyond the myometrial wall and into the isthmic section of each fallopian tube.
When one understands the outer coil is more visible from the outside of the fallopian tube and the inner coil is less visible and projects beyond the more easily visible outer coil then it is easier to understand how transection across the micro-insert could occur when removing Essure during salpingectomy or when hysterectomy is performed without salpingectomy.
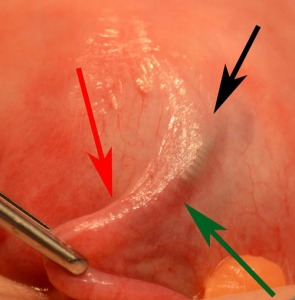
Black arrow shows easily visible external coil.
Green arrow is the approximate area where the outer coil is present but less less visible as it transitions in size to rejoin the inner coil.
Red arrow is the approximate location of the less visible distal tip of the inner coil.
Advice for removing Essure
Some of our patients with Essure micro-inserts have thickened tubal muscularis in the immediate location of the Essure devices. When this occurs direct visualization of the Essure device becomes even more difficult.
We have had good results locating the lateral most portion of the inner coil using digital palpation or by utilizing the tactile feedback provided when grabbing the micro-inserts with Debakey forceps.
If we are uncertain of the location of the lateral most tip of the micro-insert, we will dissect into the isthmic section of the fallopian tube using sharp dissection to confirm we are beyond the lateral edge of the intratubal micro-insert.
We avoid electrodissection when removing Essure because we have observed the outer coil to burn, become more brittle, and fragment more easily after exposure to electric current.
Challenges of Removing Essure: Device location is important
When the Essure devices are not in the correct location removing Essure without fragmentation can be even more challenging.
Essure coils may not be inserted far enough (shallow insertion) into the fallopian tube or they can be inserted too far (deep insertion) into the fallopian tube.
Shallow Essure insertion
When the coils are not inserted far enough removing Essure with an abdominal approach may be more challenging. The distal tip of the device may not be visible. If the distal tip can be located after a shallow coil insertion, traction during removal may increase the risk of coil fragmentation because more coil has to be pulled through dense fibroelastic tissue. This may result in micro-insert fragmentation.
Deep Essure insertion
When the coils are inserted too far into the fallopian tube the majority of the coil will be located in the isthmic or ampullary section of the fallopian tube. Essure devices which have been deeply inserted may be at increased risk for transection when removing Essure during a vaginal hysterectomy or during an abdominal hysterectomy when performed without salpingectomy.
It is also important to acknowledge Essure device placement may occur according to the manufacturer’s recommendation, be in the correct location, but the coils can migrate soon after insertion. This migration may result in the coils being more, or completely, intrauterine or extrauterine.
In our experience we have performed Essure device removal when the coils were completely expelled into the uterine cavity with persistent tubal patency, mostly in the uterine cavity while causing tubal occlusion, partially in the proximal most interstitial portion of the tube with myometrial perforation and tubal occlusion, and mostly in the isthmic section of the fallopian tube.
Expecting different degrees of micro-insert projection into and beyond the interstitial portion of the fallopian tube is important to avoid Essure fragmentation during removal.
Removing Essure after an Essure complication
This photo illustrates the challenges that should be expected when removing Essure coils intact when the coils are in correct and incorrect locations.
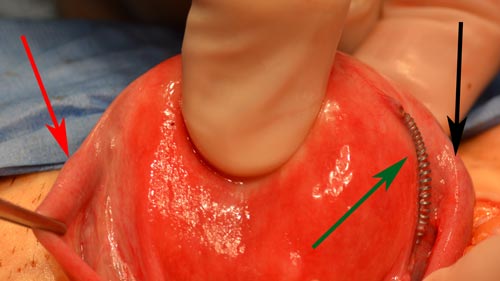 The green arrow demonstrates an ectopic Essure coil which is perforating the cornual myometrium.
The green arrow demonstrates an ectopic Essure coil which is perforating the cornual myometrium.
The device is partially in the interstitial portion of the right fallopian tube but exits the myometrium and is mostly extra-uterine.
The black arrow demonstrates the right fallopian tube.
The red arrow demonstrates the left fallopian tube. The left Essure intratubal micro-insert is in the correct location; however, the reactive thickened tubal muscularis prevents external visualization of the micro-insert. The left device was not visible but could be palpated. The thickened tubal muscularis prevented palpation of the finer distal tip of the device.
The ectopic Essure coil should be easier to remove because most of the coil is visible; however, direct traction on this coil risks fracturing the proximal most tip of the device inside the myometrium. The left Essure device could easily be transected during salpingectomy or during hysterectomy without salpingectomy because the isthmic portion of the micro-insert device is not easily visible.

