Risks of Removing Essure During Surgery
Essure removal With Dr Monteith: Analysis of 469 Patients
From January of 2009 to January of 2018, Dr. Monteith performed 469 surgeries to remove Essure sterilization devices.
This group of patients was comprised of women who requested Essure reversal (procedure to remove Essure and provide the chance of becoming pregnant) and also women who requested Essure removal (procedure to remove Essure devices for symptom treatment but prevents the chance of pregnancy).
The following statistics will refer to both groups of patients using the term Essure removal.
The following intra-operative complications were observed in the group of 469 patients who had Essure removal surgery with Dr. Monteith.
Failure to Remove Both Essure Devices <1%
In some patients it may not be possible to remove one or both Essure devices.
Three (3) patients could not have both their Essure devices removed.
Patient #1. Patient was obese (BMI>30) and had severe scar tissue as a result of several prior cesarean deliveries. One device was removed successfully. The other device (including the fallopian tube and ovary) could not be visualized due to extensive scar tissue involving the uterus and the intestines. The patient was advised to consider a hysterectomy because of the severity of her adhesions.
More information: Severe scar tissue after C-section
Patient #2. Patient was able to have one Essure device removed and bilateral tubouterine implantation (both tubes reimplanted into the uterus). One Essure device had migrated from the fallopian tube and was deeply embedded in the wall of her sigmoid colon.
Removal of the migrated device from the wall of the colon was not safe in an outpatient setting. If the migrated device was not removed properly the patient could have suffered severe complications and may have required a temporary colostomy.
The patient was advised to attempt pregnancy and complete all desired pregnancy goals. Removal of the migrated Essure device could be done in the future but would require a colorectal surgeon, major surgery in a hospital setting with proper preoperative bowel preparation, and removal of part of her colon.
Patient #3. Patient had prolonged bradycardia (low heart rate) at the start of the Essure removal procedure. Neither Essure device was removed. She was transferred to the hospital for observation. See hospital transfer below for further details.
Failure to complete tubouterine implantation (Essure reversal) <1%
In some patients Essure devices can be removed but tubouterine implantation (Essure reversal) may not be completed. Two (2) patients could not have bilateral tubouterine implantation performed.
Patient #1. Patient had a history of 3 prior cesarean deliveries, obesity (BMI 30), and had uterine fibroids. Both Essure devices were removed. Due to increased bleeding from the presence of uterine fibroids, bilateral tubouterine implantation was not performed. The patient did well after surgery and recovered without complications.
Patient #2. Patient was obese (BMI 35) and had a history of a Cesarean delivery and abdominoplasty (tummy tuck). During Essure reversal, uterine fibroids were encountered. Both Essure devices were removed. Due to increased bleeding from the presence of uterine fibroids, bilateral tubouterine implantation was not performed. The patient did well after surgery and recovered without complications.
Fracture of Essure devices: <1% with En Bloc Dissection Technique

The outer coil is made of a nickel alloy (nitinol) with a rectangular marker (platinum band) on the end of the outer coil. The inner coil is made of stainless steel intertwined with PET fibers.
The outer nitinol coil is more prone to fracture (especially if electrocautery is used).
The rectangular marker (platinum band) is welded to the nitinol coil and this weld is a weak point and prone to fracture/separate if manual traction is used.
The inner stainless steel coil is extremely strong and less prone to fracture.
The device is prone to fracture under the following circumstances:
- When manual traction is applied
- When electrocoagulation is used
It is difficult to provide a precise estimate of the risk of Essure device fracture during removal surgery.
The surgeon has to have the experience and skill to suspect and detect a fractured device at the time of the procedure. The surgeon also has to have the willingness to divulge the possibility of device fracture to the patient. Sometimes device fracture is not detected until well after surgery has been completed and fragments are visualized on x-ray.
Prior to 2014, we estimated the chance of Essure device fracture to be approximately 10%. This estimate was based on clinical suspicion based on the appearance of the devices at the time of manual extraction.
Prior to 2014, we utilized a manual traction technique to remove Essure devices (we pulled on the devices). We did encounter fracture of the devices. Sometimes this was recognized at the time of surgery and the fragments were retrieved. Sometimes the fragments could not be retrieved. Sometimes fracture was not recognized until pregnancy did not occur and patients obtained HSG x-rays to determine if their fallopian tubes were open.
More info: Manual traction to remove Essure devices
In 2014, we stopped using manual traction and switched to an en bloc complete dissection of the Essure devices without using electrocoagulation.
After 2014, we estimated the chance of Essure device fracture to be <1% based on clinical suspicion at the time of surgery.
More info: En bloc dissection of Essure devices
Since we have been performing en bloc dissections to remove the Essure devices, we believe our risk of device fragmentation is much lower. Using our en bloc dissection technique, most Essure devices can be dissected completely, without traction, and without using electrocoagulation.
If Essure removal is difficult because of dense adhesions, uterine fibroids, or increased bleeding, then we will remove devices with manual traction and the chance of device fragmentation is increased when this technique is employed.
Bleeding Requiring Blood Transfusion
Bleeding requiring hospitalization or blood transfusion 0%
The average estimated blood loss for our Essure removal/reversal surgery is 75 milliliters (the equivalent to five (5) tablespoons).
Essure removal has the potential to be a bloody procedure because of the large amount of blood flow to the uterus. None of our patients experienced heavy bleeding during surgery requiring transfer to a hospital or a blood transfusion.
Blood flow to the uterus is increased significantly in women who have large (>5 cm) uterine fibroids. Women who have large uterine fibroids are at increased risk of not having a tubouterine implantation completed and Essure device fragmentation.
If Essure removal patients experience increased bleeding during surgery we will abandon an en bloc dissection because of the increased chance of hemorrhage. Instead we will remove the devices with manual traction even though this procedure increases the risk of device fragmentation. The risk of heavy bleeding is less with manual extraction of the devices.
If bleeding is excessive then a tubouterine implantation will not be performed because of the potential of increased bleeding during the implantation portion of the procedure.
Anesthesia Complications 0%
None of our patients experienced an anesthesia complication.
Hospital Transfer During Surgery or Hospital/ER Referral Within 24 hours of surgery <1%
Patient #1. This patient was mentioned above. During the start of device removal, the patient experienced bradycardia (low heart rate). The surgery was discontinued and the patient was transferred to the hospital. She was admitted for observation. She was ultimately discharged home without any significant complication. Her bradycardia likely resulted from manipulation of the uterus during surgery. This commonly happens during cesarean delivery.
Patient #2. One Essure reversal patient experienced dizziness and a fainting episode the night of surgery. She was evaluated in the Emergency Room by Dr. Monteith, no cause could be determined, and she was allowed to return to the hotel to rest. She returned for her post-operative exam with Dr. Monteith the next day and had fully recovered. The patient did well after surgery and recovered without complications.
Death 0%
Death is the ultimate adverse outcome of surgery. To date, no patient has died as a direct result of Essure removal surgery at our center.
Risks of Removing Essure: 3 Factors That Increase Risks of Removal
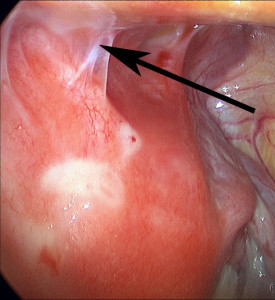
Arrow demonstrates uterine adhesion to abdominal muscles in patient with a history of C-sections
Three factors clearly increase the risks of removing Essure: (1) uterine and intestinal adhesions (scar tissue), (2) uterine fibroids, (3) patient obesity.
Adhesions
The presence of severe scar tissue makes Essure removal more difficult and increases the risks of removing Essure. Sometimes manual traction will need to be performed and this increases the risk of device fragmentation. Sometimes severe scar tissue will prevent visualization of a Essure device in the tube and removal may not be possible.
The risk of having severe scar tissue is increased in the following situations:
- History of cesarean
- History of Classical cesarean (vertical incision on both abdomen and uterus)
- Abdominal surgery to remove multiple large uterine fibroids
- Intestinal surgery complicated by infection
Uterine Fibroids
Uterine fibroids can be very common. The chance of having uterine fibroids is higher in African American women. The presence of large (> 5cm) uterine fibroids can increase the risks of removing Essure and increase the risk of having heavy bleeding during surgery. If bleeding is heavy then both removal of devices and tubouterine implantation may not be possible.
Obesity
Patient obesity (BMI >30) makes surgery more difficult. In our experience, obesity alone is not a strong risk factor for increasing the risks of removing Essure during surgery; however, obesity combined with the presence of severe adhesions and/or large uterine fibroids greatly increases the difficulty of removing surgery.

